CLINICAL EFFICACY
Post hoc analysis: NEDA (no evidence of disease activity) and efficacy in different subgroups and overall study population in patients treated with MAVENCLAD versus placebo1
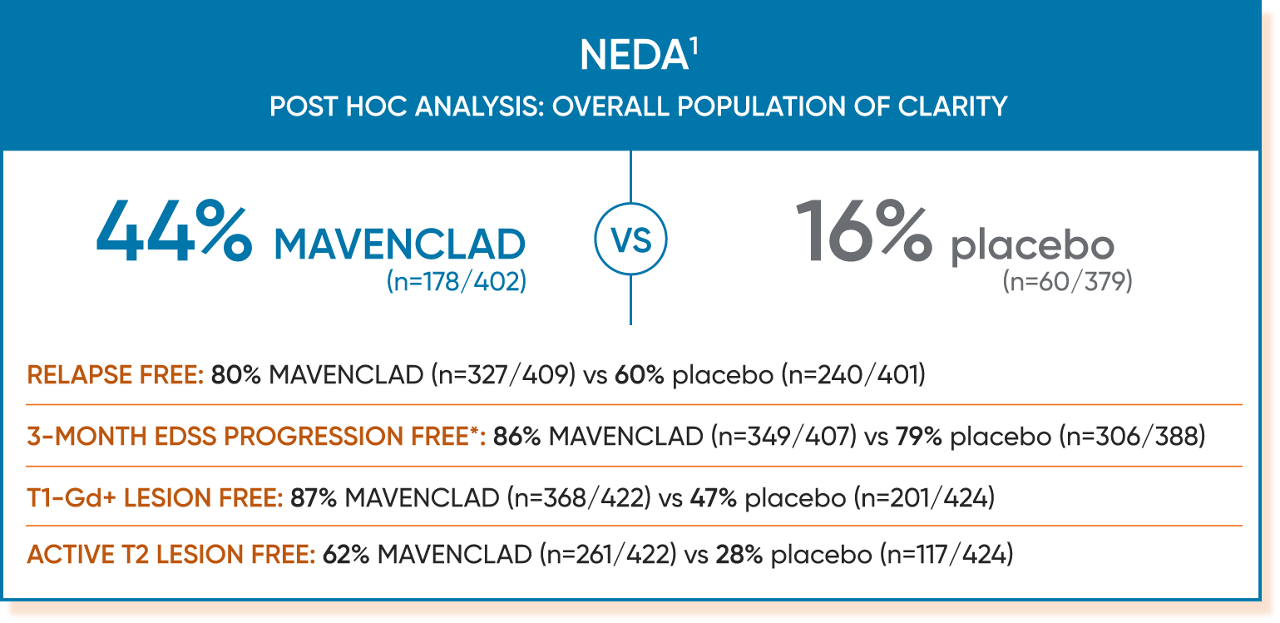
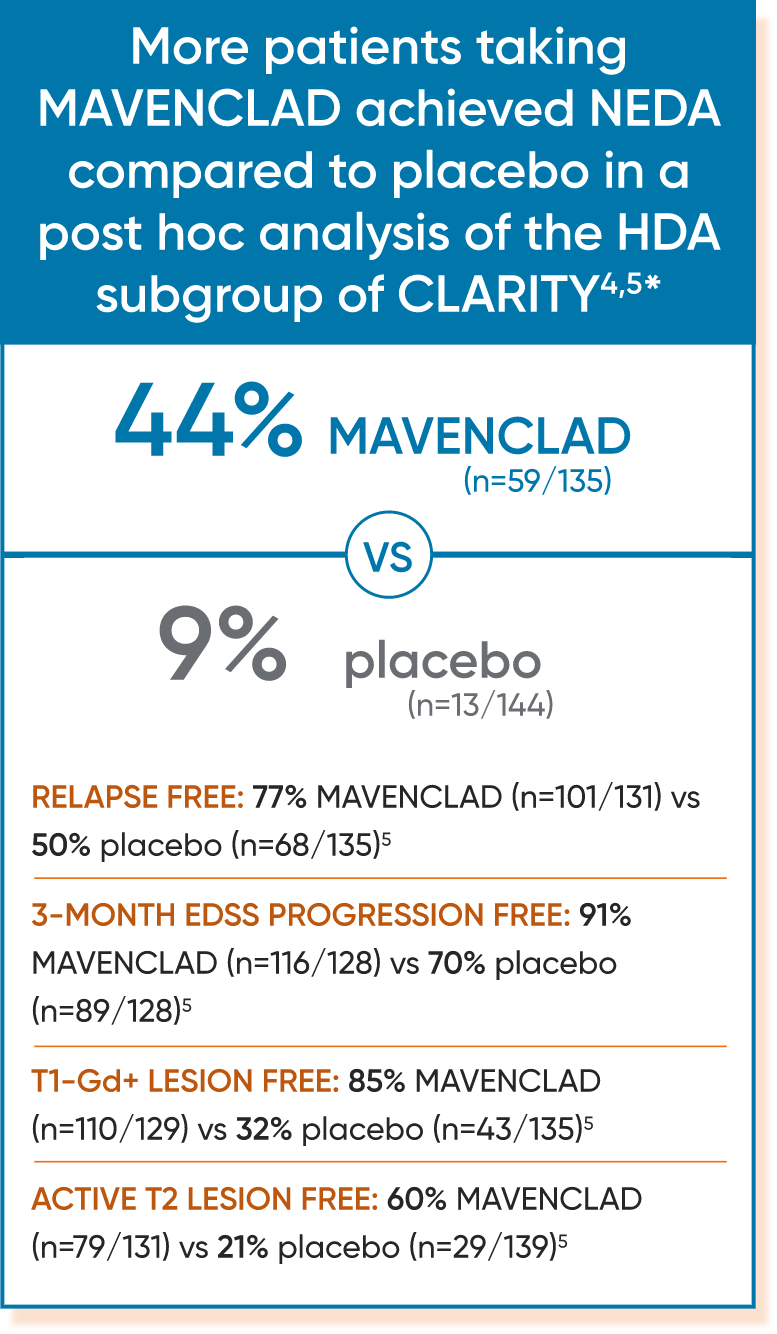
In a post hoc analysis of CLARITY, more patients taking MAVENCLAD achieved NEDA compared to placebo.1

Limitations of composite endpoints include assuming that components have equal weight and are independent of one another—which is not necessarily the case.1
Due to the post hoc nature of this analysis, no statistical significance between treatment groups can be drawn.
Of the 870 patients in the CLARITY study who received MAVENCLAD 3.5 mg/kg or placebo, 781 were assessable for NEDA at 96 weeks. MRI scans were obtained at the prestudy evaluation and at 24, 48, and 96 weeks.1
NEDA was defined in CLARITY as patients who1:
*The 3-month EDSS progression–free component of the NEDA analysis included the 407 patients in the MAVENCLAD arm and 388 patients in the placebo arm who were assessable for NEDA.1
†A relapse was defined as a 2-point increase in ≥1 functional system of the EDSS or a 1-point increase in ≥2 functional systems (excluding changes in bowel or bladder function or cognition) in the absence of fever, lasting for ≥24 hours and preceded by ≥30 days of clinical stability or improvement.1
‡Disability progression was measured in terms of a 3-month sustained change in EDSS score of at least 1 point, if the baseline EDSS score was between 0.5 and 4.5 inclusively, or at least 1.5 points if the baseline EDSS score was 0, or at least 0.5 point if the baseline EDSS score was at least 5, over a period of at least 3 months.2
NEDA: free from both clinical (relapse and EDSS) and radiological (MRI) activity.
CLARITY: CLAdRIbine Tablets treating multiple sclerosis orallY; EDSS: Expanded Disability Status Scale; NEDA: no evidence of disease activity; T1-Gd+: T1 gadolinium-enhanced.
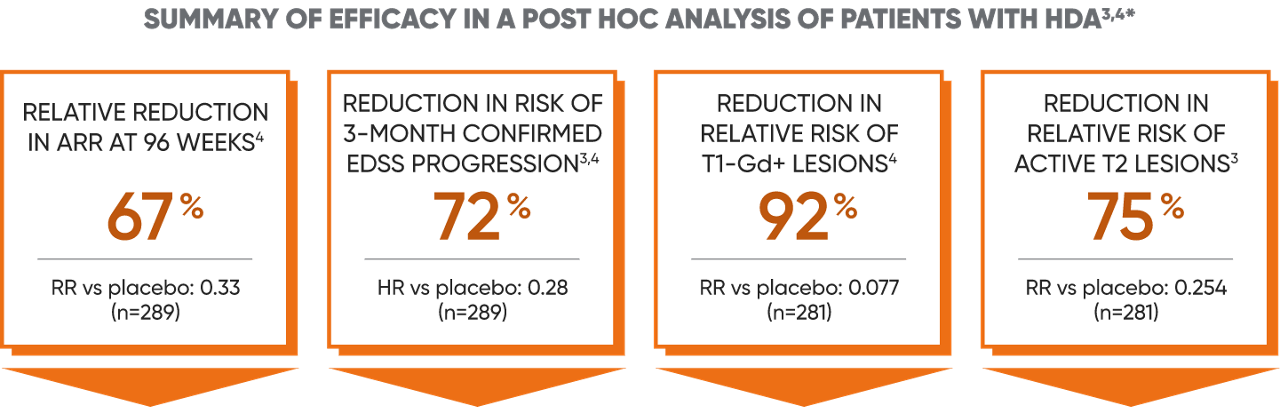
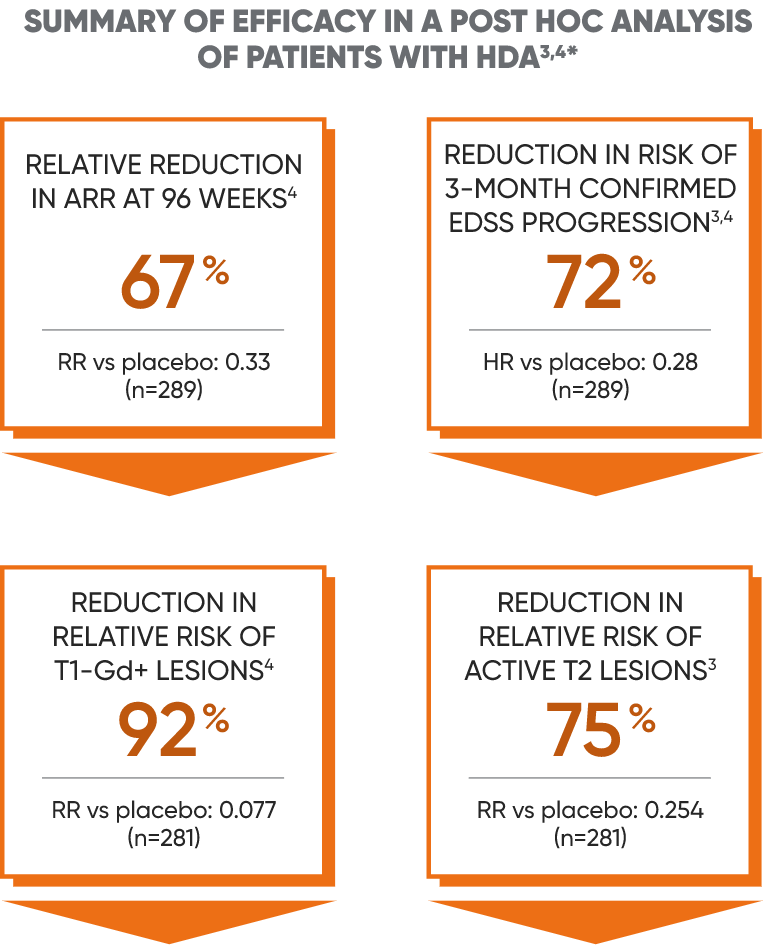
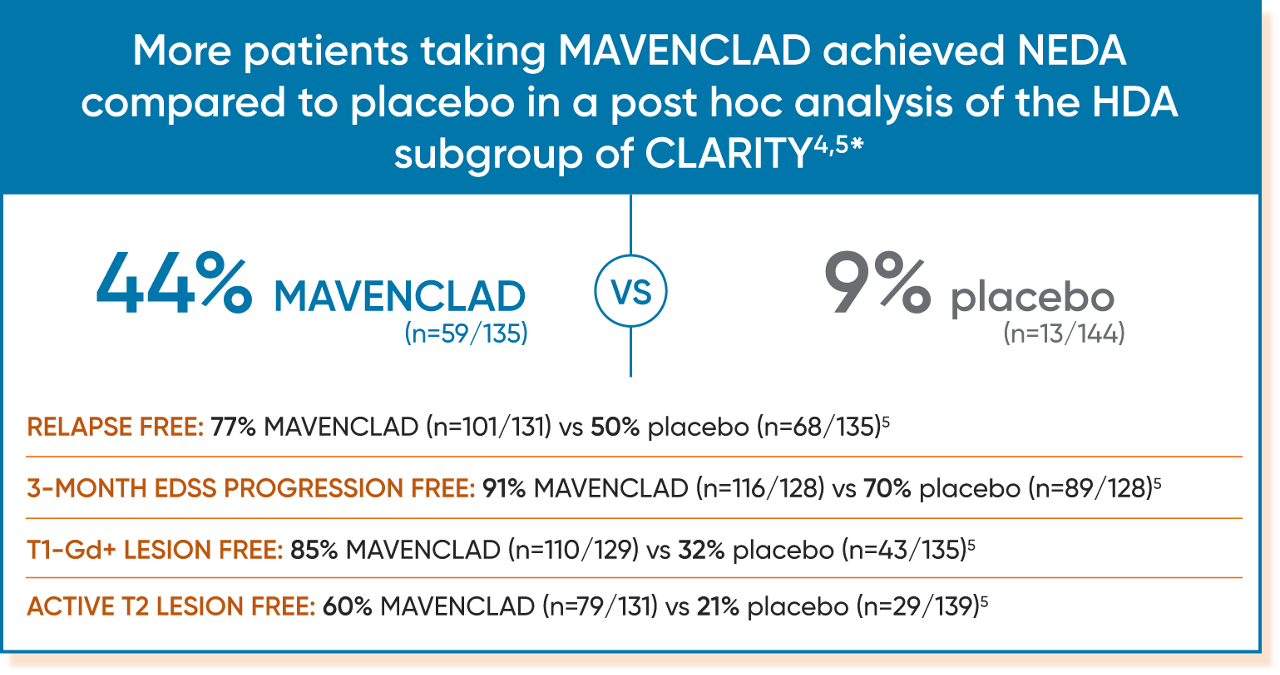
Post hoc analysis: Efficacy in a subgroup of patients with HDA (high disease activity)3,4
Disease activity in patients with relapsing MS can be predictive of future disease worsening and overall poor prognosis.4
Subgroup of patients in CLARITY who had4:
- ≥2 relapses in the year before study entry, regardless of treatment status, OR
- ≥1 relapse in the year before study entry while on another DMT AND ≥1 T1-Gd+ or ≥9 T2 lesions
The HDA subgroup did not reveal evidence for new safety findings compared with those described for the overall CLARITY population.4
Patients had a mean disease duration of:5
- 4.75 years for placebo (standard deviation: 5.43)
- 3.94 years for MAVENCLAD (standard deviation: 4.92)
- Patients with HDA compared to the overall study population had a higher number of relapses during the 12 months prior to baseline
Limitations of composite endpoints include assuming that components have equal weight and are independent of one another—which is not necessarily the case.1
Due to the post hoc nature of this analysis, no statistical significance between treatment groups can be drawn.
*Data were derived from a post hoc analysis of CLARITY. Qualifying relapses were defined as a 2-grade increase in 1 or more KFS or a 1-grade increase in 2 or more KFS, excluding changes in bowel/bladder or cognition, in the absence of fever, lasting for ≥24 hours, and preceded by ≥30 days of clinical stability or improvement. Disability progression was measured in terms of a 3-month sustained change in EDSS score of at least 1 point, if baseline EDSS score was between 0.5 and 4.5 inclusively, or at least 1.5 points if the baseline EDSS score was 0, or at least 0.5 point if the baseline EDSS score was at least 5, over a period of at least 3 months. P value for imaging measurement was based on a nonparametric analysis-of-covariance model on ranked data with fixed effects for study group and region and the number of T1-Gd+–weighted lesions at baseline as a covariate.4,6
ARR: annualized relapse rate; CLARITY: CLAdRIbine Tablets treating multiple sclerosis orallY; DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; HDA: high disease activity; HR: hazard ratio; KFS: Kurtzke functional score; NEDA: no evidence of disease activity; RR: relative risk.
Post hoc analysis: High EDSS, a proxy for active SPMS7
Secondary progressive multiple sclerosis (SPMS)
SPMS is a process characterized by progression of disability independent of relapses.8
Active SPMS
In the first few years of this process, many patients continue to experience relapses—a phase of the disease described as active SPMS.8,9 Active SPMS is one of the relapsing forms of MS (RMS).9,10
Non-active SPMS
Many patients with SPMS stop experiencing relapses, but disability continues to progress—a phase called non-active SPMS.9
EDSS score as a proxy for active SPMS
One proposed definition for active SPMS is that RMS patients with an EDSS of ≥3.5 can serve as a proxy of active SPMS.7,8,10,11
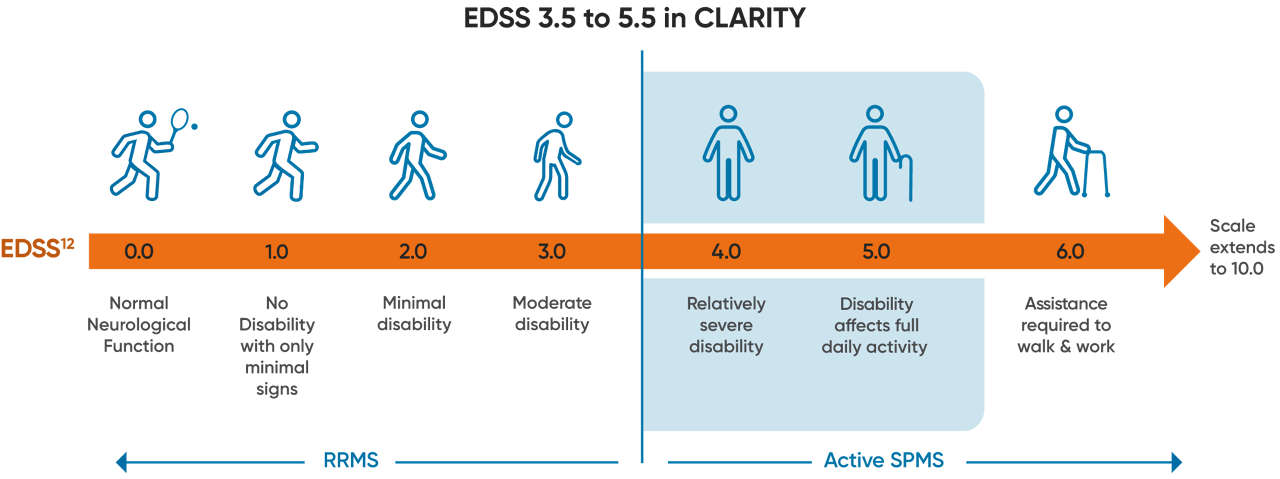

Active SPMS subgroup in CLARITY
Approximately 40% (n=335/870) of MAVENCLAD and placebo patients in the CLARITY trial met the active SPMS proxy criteria of EDSS ≥3.5 (all patients in CLARITY were required to have at least 1 relapse in the previous 12 months) and EDSS score ≤5.5.2,13
CLARITY: CLAdRIbine tablets treating multiple sclerosis orallY; EDSS: Expanded Disability Status Scale; RRMS: relapsing-remitting multiple sclerosis.
Post hoc analysis: MAVENCLAD reduced ARR vs placebo in active SPMS13
In a post hoc analysis of CLARITY, MAVENCLAD reduced the ARR vs placebo in patients who met the proxy criteria for active SPMS.13
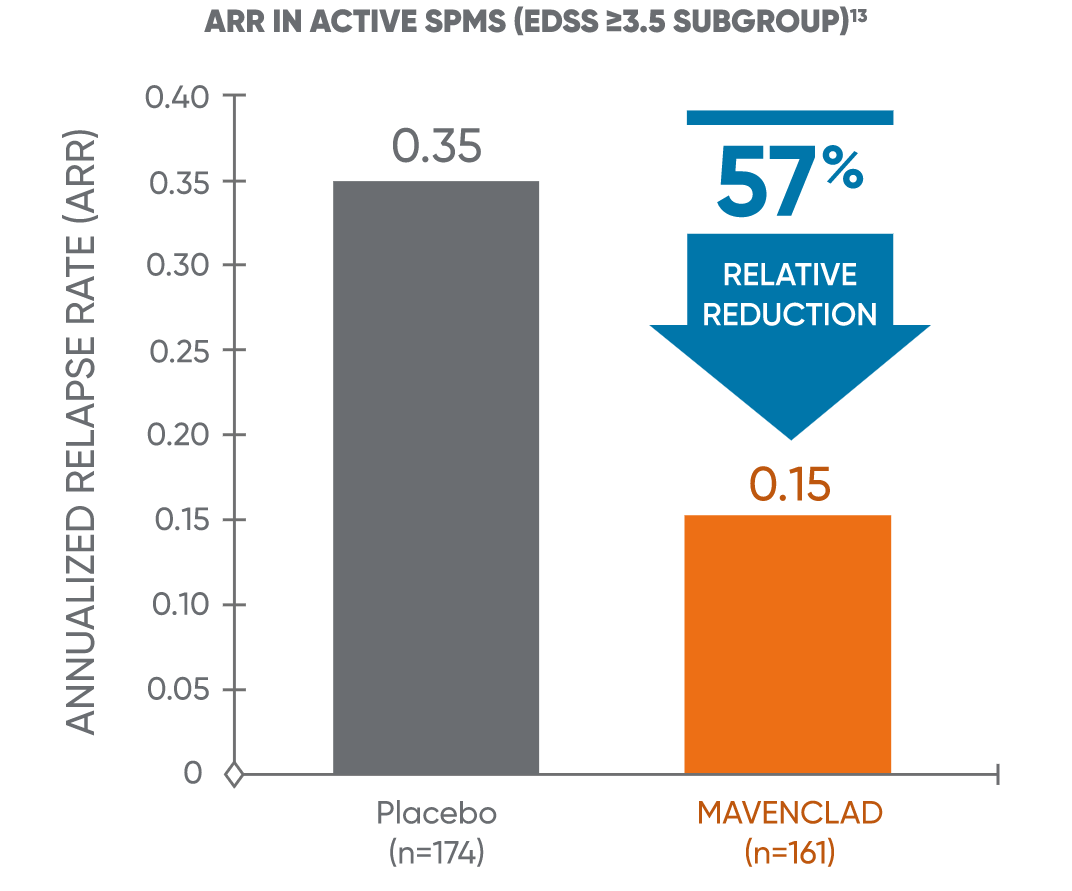
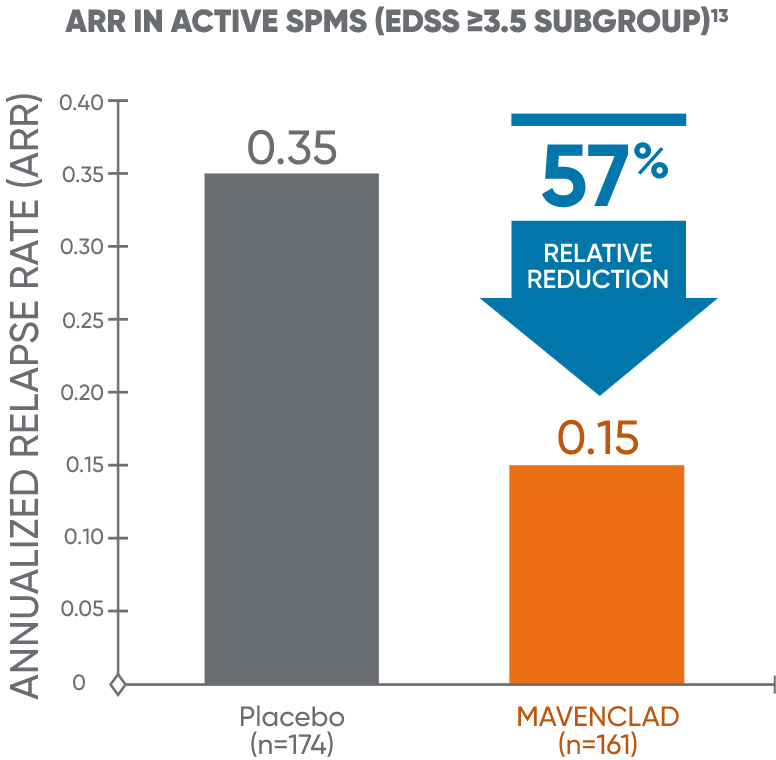
ARR: annualized relapse rate; CLARITY: CLAdRIbine tablets treating multiple sclerosis orallY; EDSS: Expanded Disability Status Scale; SPMS: secondary progressive multiple sclerosis.
Post hoc analysis of CLARITY: MAVENCLAD patients with active SPMS vs placebo1*
More patients who met the proxy criteria for active SPMS achieved NEDA with MAVENCLAD vs placebo.1*

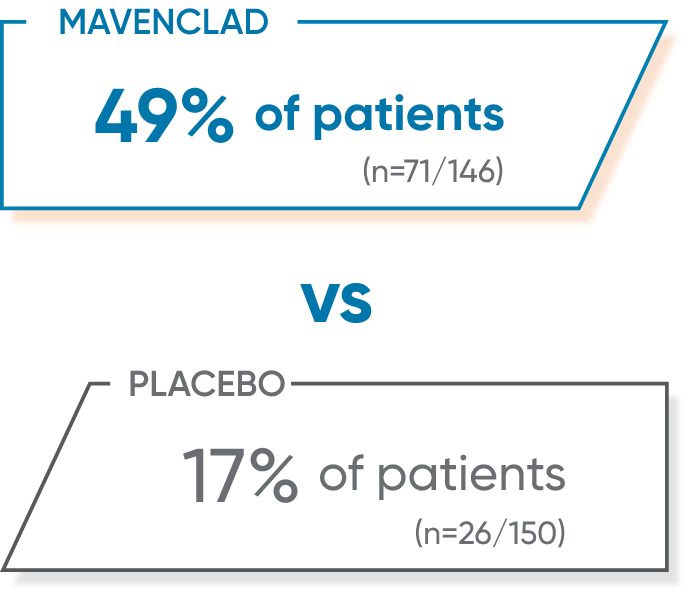
Limitations of composite endpoints include assuming that components have equal weight and are independent of one another—which is not necessarily the case.1
Due to the post hoc nature of this analysis, no statistical significance between treatment groups can be drawn.
*The definition of NEDA is consistent with that used for the overall population as outlined above.
CLARITY: CLAdRIbine tablets treating multiple sclerosis orallY; NEDA: no evidence of disease activity; SPMS: secondary progressive multiple sclerosis.
INDICATION and IMPORTANT SAFETY INFORMATION for MAVENCLAD® (cladribine) tablets
MAVENCLAD® (cladribine) is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include relapsing-remitting disease and active secondary progressive disease, in adults. Because of its safety profile, use of MAVENCLAD is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS.
Limitations of Use: MAVENCLAD is not recommended for use in patients with clinically isolated syndrome (CIS) because of its safety profile.
IMPORTANT SAFETY INFORMATION
WARNING: MALIGNANCIES and RISK OF TERATOGENICITY
- Treatment with MAVENCLAD may increase the risk of malignancy. MAVENCLAD is contraindicated in patients with current malignancy. In patients with prior malignancy or with increased risk of malignancy, evaluate the benefits and risks of the use of MAVENCLAD on an individual patient basis. Follow standard cancer screening guidelines in patients treated with MAVENCLAD.
- MAVENCLAD is contraindicated for use in pregnant women and in women and men of reproductive potential who do not plan to use effective contraception because of the potential for fetal harm. Malformations and embryolethality occurred in animals. Exclude pregnancy before the start of treatment with MAVENCLAD in females of reproductive potential. Advise females and males of reproductive potential to use effective contraception during MAVENCLAD dosing and for 6 months after the last dose in each treatment course. Stop MAVENCLAD if the patient becomes pregnant.
CONTRAINDICATIONS
- Patients with current malignancy.
- Pregnant women, and women and men of reproductive potential who do not plan to use effective contraception during and for 6 months after the last dose in each treatment course. May cause fetal harm.
- Patients infected with human immunodeficiency virus (HIV).
- Patients with active chronic infections (e.g., hepatitis or tuberculosis).
- Patients with a history of hypersensitivity to cladribine.
- Women intending to breastfeed on a MAVENCLAD treatment day and for 10 days after the last dose.
WARNINGS AND PRECAUTIONS
- Malignancies: Treatment with MAVENCLAD may increase the risk of malignancy. After the completion of 2 treatment courses, do not administer additional MAVENCLAD treatment during the next 2 years. In clinical studies, patients who received additional MAVENCLAD treatment within 2 years after the first 2 treatment courses had an increased incidence of malignancy. The risk of malignancy with reinitiating MAVENCLAD more than 2 years after the completion of 2 treatment courses has not been studied. Follow standard cancer screening guidelines in patients treated with MAVENCLAD.
- Risk of Teratogenicity: MAVENCLAD may cause fetal harm when administered to pregnant women. In females of reproductive potential, exclude pregnancy before initiation of each treatment course of MAVENCLAD and prevent by the use of effective contraception during MAVENCLAD dosing and for at least 6 months after the last dose of each treatment course. Women who become pregnant during treatment with MAVENCLAD should discontinue treatment.
- Lymphopenia: MAVENCLAD causes a dose-dependent reduction in lymphocyte count. Concomitant use of MAVENCLAD with hematotoxic drugs may increase the risk of adverse reactions because of the additive hematological effects. Monitor lymphocyte counts before, during, and after treatment.
- Infections: Serious, including life-threatening or fatal, infections have occurred. MAVENCLAD reduces the body's immune defense, and an increased risk of infections has been observed in patients receiving MAVENCLAD. Infections occurred in 49% of MAVENCLAD-treated patients compared to 44% of patients treated with placebo in clinical studies; serious or severe infections occurred in 2.4% of MAVENCLAD- treated patients and 2.0% of placebo-treated patients. The most frequent serious infections included herpes zoster and pyelonephritis. Fungal infections were observed, including cases of coccidioidomycosis. Single fatal cases of tuberculosis and fulminant hepatitis B were reported in the clinical program.
- Screen patients for active and latent infections (tuberculosis, hepatitis B or C). Delay treatment until infection is fully resolved or controlled.
- Vaccinate patients who are seronegative for varicella zoster virus (VZV) prior to treatment. Vaccinate patients who are seropositive to VZV with recombinant, adjuvanted zoster vaccine either prior to or during treatment, including when their lymphocyte counts are less than or equal to 500 cells per microliter.
- Administer anti-herpes prophylaxis in patients with lymphocyte counts less than 200 cells per microliter. Monitor for infections.
- Progressive multifocal leukoencephalopathy (PML) has been reported in patients treated with parenteral cladribine for oncologic indications. No case of PML has been reported in clinical studies of cladribine in patients with MS. Obtain a baseline magnetic resonance imaging (MRI) within 3 months before initiating the first treatment course of MAVENCLAD. At the first sign of PML, withhold MAVENCLAD and perform an evaluation.
- Administer all immunizations (except as noted for VZV) according to immunization guidelines prior to starting MAVENCLAD. Administer live-attenuated or live vaccines at least 4 to 6 weeks prior to starting MAVENCLAD due to risk of infection.
- Hematologic Toxicity: In addition to lymphopenia, decreases in other blood cells and hematological parameters have been reported with MAVENCLAD in clinical studies. Obtain complete blood count (CBC) with differential including lymphocyte count before and during treatment, periodically thereafter, and when clinically indicated.
- Graft-versus-Host Disease with Blood Transfusions: Transfusion-associated graft-versus-host disease has been observed rarely after transfusion of nonirradiated blood in patients treated with cladribine for non-MS treatment indications. In patients who require blood transfusion, irradiation of cellular blood components is recommended.
- Liver Injury: In clinical studies, 0.3% of MAVENCLAD-treated patients had liver injury (serious or causing treatment discontinuation) compared to 0 placebo patients. Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels prior to treatment. Discontinue MAVENCLAD if clinically significant liver injury is suspected.
- Hypersensitivity: If a hypersensitivity reaction is suspected, discontinue MAVENCLAD therapy. Do not use MAVENCLAD in patients with a history of hypersensitivity to cladribine.
- Cardiac Failure: In clinical studies, one MAVENCLAD-treated patient experienced life-threatening acute cardiac failure with myocarditis, which improved after approximately one week. Cases of cardiac failure have also been reported with parenteral cladribine used for treatment indications other than multiple sclerosis. Instruct patients to seek medical advice if they experience symptoms of cardiac failure (e.g., shortness of breath, rapid or irregular heartbeat, swelling).
Adverse Reactions: The most common adverse reactions (incidence of >20%) are upper respiratory tract infection, headache, and lymphopenia.
Drug Interactions: Concomitant use with immunosuppressive or myelosuppressive drugs and some immunomodulatory drugs (e.g., interferon beta) is not recommended and may increase the risk of adverse reactions. Acute short-term therapy with corticosteroids can be administered. Monitor for additive effects on the hematological profile with use of hemotoxic drugs. Avoid concomitant use of antiviral and antiretroviral drugs. Avoid concomitant use of BCRP or ENT/CNT inhibitors as they may alter bioavailability of MAVENCLAD.
Use in Specific Populations: Studies have not been performed in pediatric, or elderly patients >65 years, pregnant or breastfeeding women. Use in patients with moderate to severe renal or hepatic impairment is not recommended.
To report SUSPECTED ADVERSE REACTIONS, contact EMD Serono, Inc. at 1-800-283-8088 ext. 5563 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see FULL PRESCRIBING INFORMATION, including BOXED WARNING.
