Malignancy rates were higher in US clinical study patients than the rest of the world (4 events in 189 patient-years [2.21 events per 100 patient-years], compared to 0 events in US placebo patients). However, the US results were based on a limited amount of patient data.1
SAFETY & TOLERABILITY
Well-characterized Safety and Tolerability Profile
The safety and tolerability of MAVENCLAD were studied in CLARITY.1
MAVENCLAD clinical experience
Over 20+ years of clinical, observational, and real-world experience in MS informs the safety profile.2
Clinical safety experience in MS
In the clinical trial program of cladribine* in MS, 1,976 patients received cladribine for a total of 9,509 patient-years.1
The mean time on study including follow-up was ~4.8 years, and ~24% of cladribine-treated patients had ~8 years of time on study including follow-up.1*
All exposed: 1,976 patients received cladribine* in the clinical program, which includes trials not depicted here.1
Monotherapy: 923 patients received MAVENCLAD† in the clinical program.1
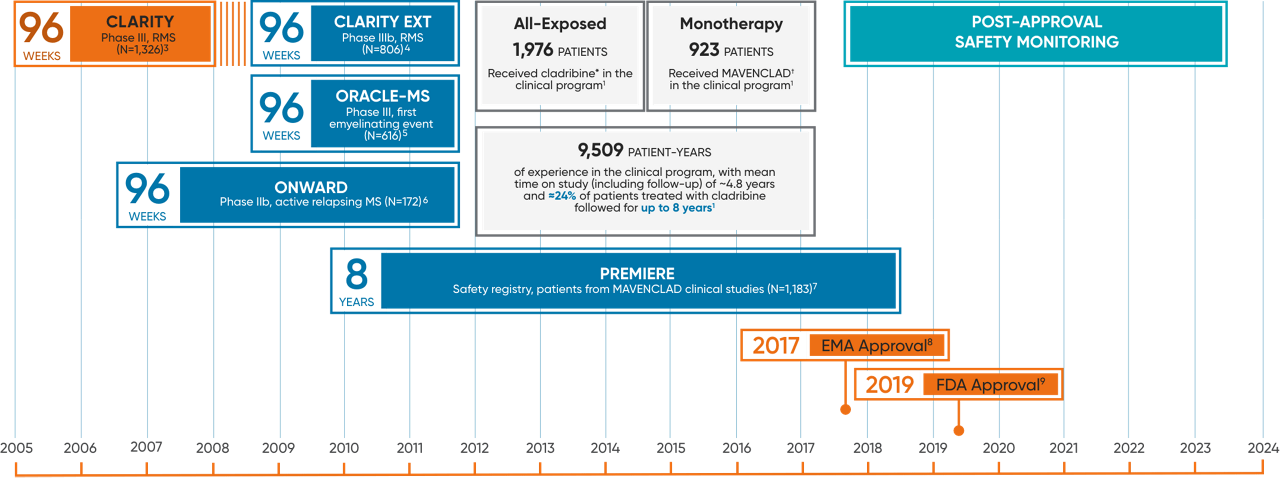
*Includes data from studies of oral and parenteral forms of cladribine.
†Includes data from the approved MAVENCLAD dose of 3.5 mg/kg over 2 years.
CLARITY: CLAdRIbine Tablets treating multiple sclerosis orallY; RRMS: relapsing-remitting multiple sclerosis.
130,000+RMS patients have been treated globally in the real-world setting for a total of330,000+patient-years10
Risk of malignancy
Treatment with MAVENCLAD may increase the risk of malignancy. MAVENCLAD is contraindicated in patients with current malignancy.1
Malignancy cases in MAVENCLAD patients occurred in several different trials. In the CLARITY MAVENCLAD group, 6 patients (1.4%) experienced malignancies compared to 0 patients in the placebo group.3
Malignancy cases in MAVENCLAD patients
Malignancy cases in MAVENCLAD patients included metastatic pancreatic carcinoma, malignant melanoma (2 cases), and ovarian cancer, compared to malignancy cases in placebo patients, all of which were curable by surgical resection (basal cell carcinoma, cervical carcinoma in situ [2 cases]).1
There were no cases of hematological or lymphoproliferative cancers in the pooled safety dataset.11
| System organ class: malignancy or unspecified tumors* | MAVENCLAD (n=10/923) | Placebo (n=3/641) |
|---|---|---|
| Basal cell carcinoma | 1 | 1 |
| Bile duct adenocarcinoma | 1 | 0 |
| Breast cancer | 1 | 0 |
| Cervix carcinoma stage 0 | 0 | 2 |
| Malignant melanoma | 2 | 0 |
| Ovarian cancer | 1 | 0 |
| Pancreatic carcinoma | 1 | 0 |
| Papillary thyroid cancer | 1 | 0 |
| Rectal cancer | 1 | 0 |
| Squamous cell carcinoma of skin | 1 | 0 |
*Malignant or unspecified tumors determined by external adjudication.
Malignancy rates in the MAVENCLAD clinical program
Treatment with MAVENCLAD may increase the risk of malignancy.1 In controlled and extension clinical studies worldwide, malignancies occurred more frequently in MAVENCLAD-treated patients (10 events in 3,754 patient-years [0.27 events per 100 patient-years]), compared to placebo patients (3 events in 2,275 patient-years [0.13 events per 100 patient-years]).1
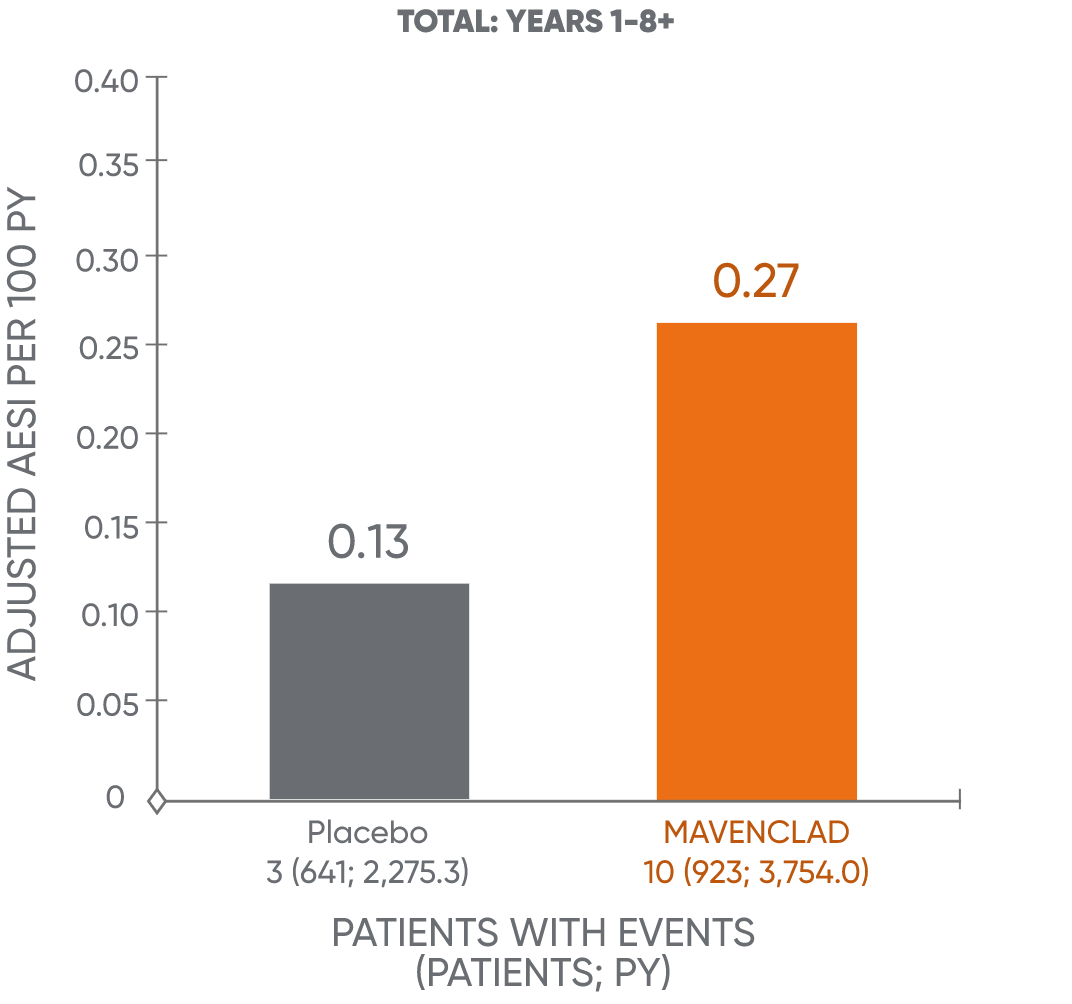
AESI: adverse events of special interest; PY: patient-years.
Malignancy rate over time
The occurrence of malignancies over time was further assessed.13 This analysis showed no additional increase over time in the adjusted malignancy incidence rate for MAVENCLAD for years 1 to 4 and years 5 to 8+.11†
Malignancy rates in years 1-4 and 5-8+11
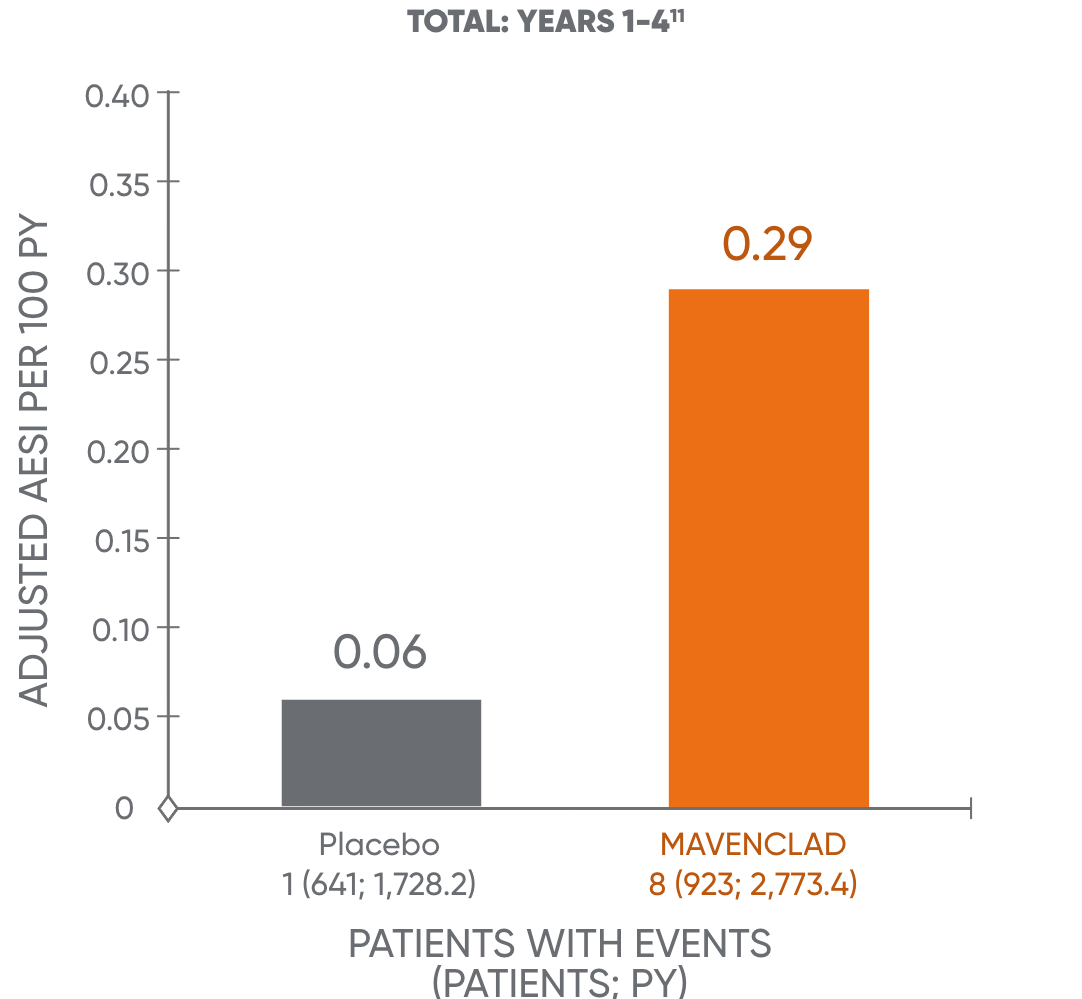
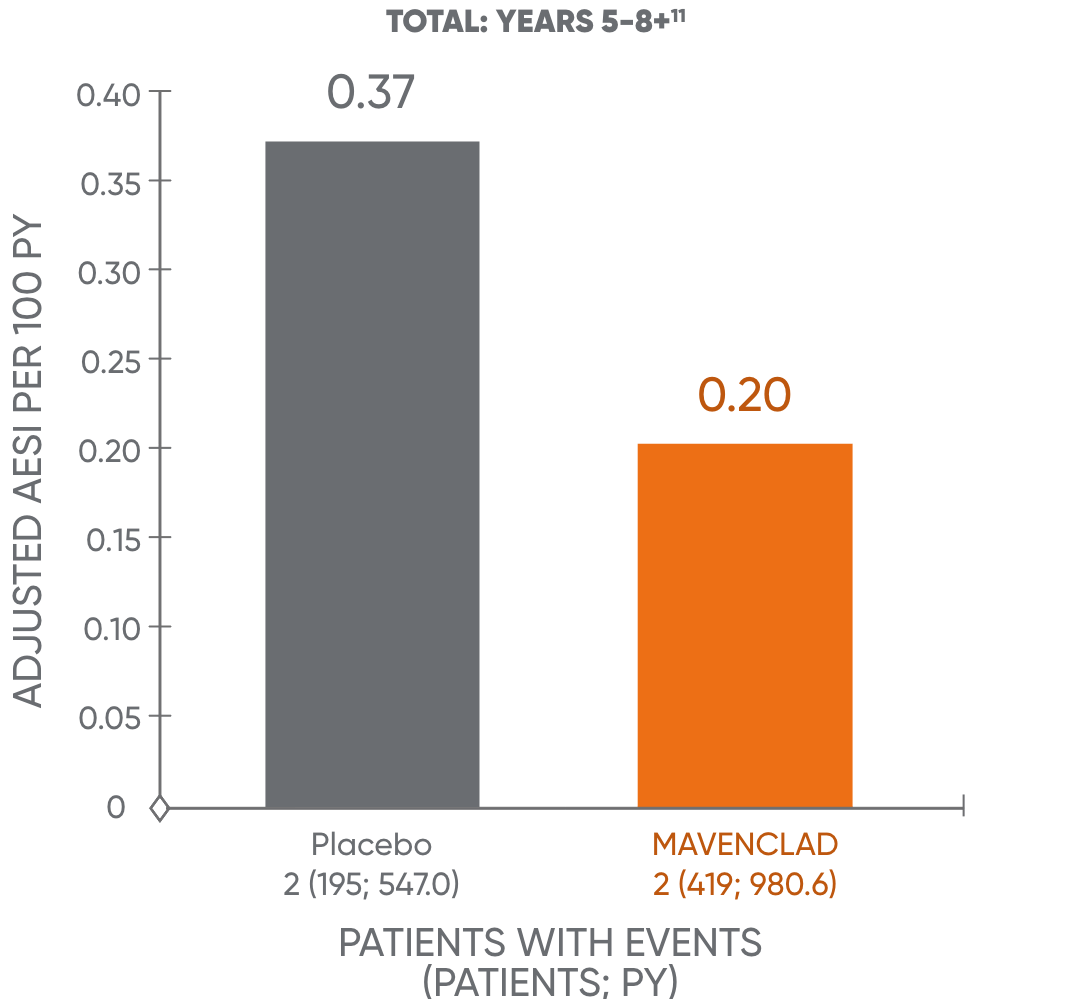
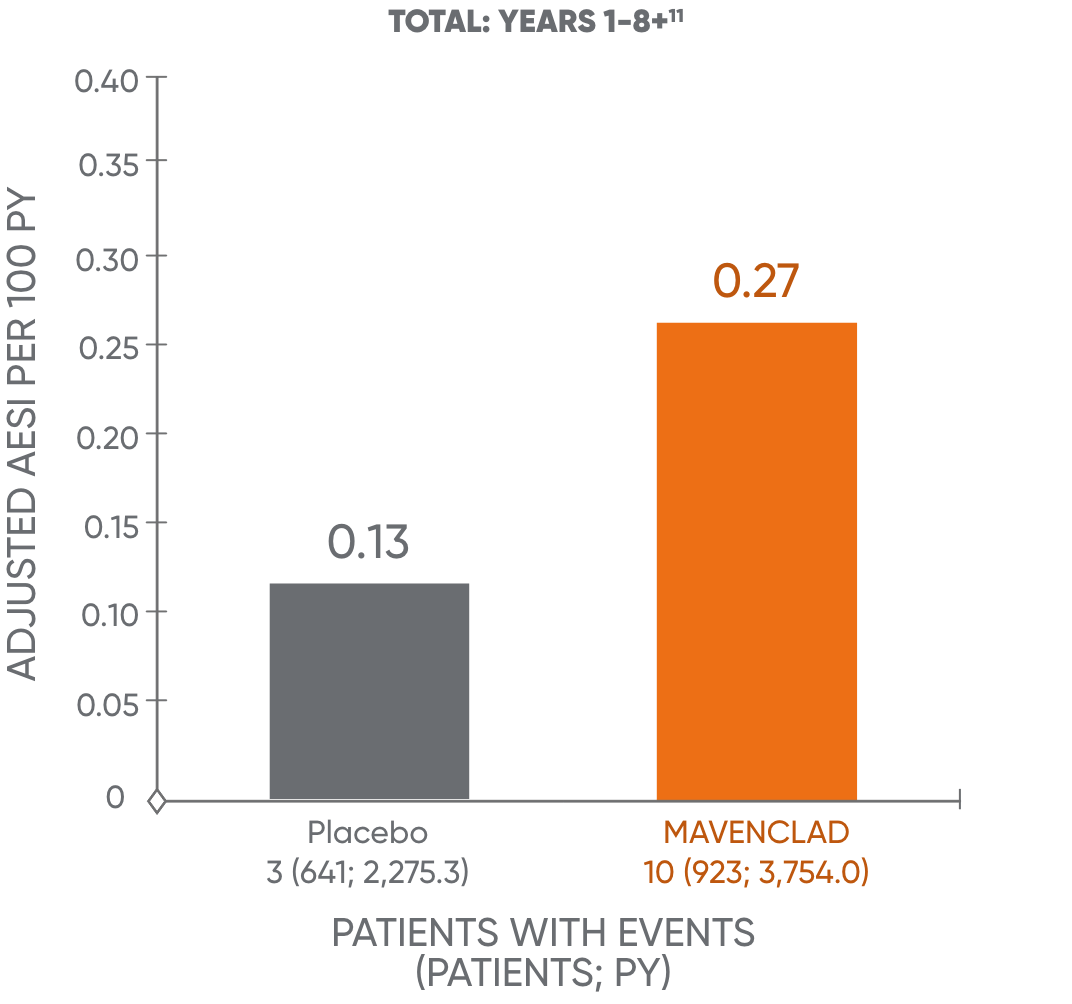
The analysis is based on a cohort of patients who were treated with MAVENCLAD over 2 years and followed without further dosing. The results of years 5 to 8+ are not dispositive and should be treated with caution. An observational study to assess the long-term risk of malignancy is being conducted.11,14
MAVENCLAD is a purine antimetabolite that interferes with DNA synthesis and repair.1
MAVENCLAD should not be used in patients with current malignancy. In MS patients with a prior malignancy, evaluate the benefits and risks on an individual patient basis. Follow standard cancer screening guidelines.1
Following the administration of 2 treatment courses, do not administer additional MAVENCLAD treatment during the next 2 years, because treatment during these 2 years may further increase the risk of malignancy.1
In clinical studies, patients who received additional MAVENCLAD treatment within 2 years after the first 2 treatment courses had an increased incidence of malignancy (7 events in 790 patient-years [0.91 events per 100 patient-years] calculated from the start of cladribine treatment in year 3). The risk of malignancy with reinitiating MAVENCLAD more than 2 years after the completion of 2 treatment courses has not been studied.1
†Company-sponsored analysis.
AESI: adverse events of special interest; PY: patient-years.
Pregnancy and teratogenicity risk1
MAVENCLAD may cause fetal harm when administered to pregnant women. MAVENCLAD is contraindicated for use in pregnant women and in women and men of reproductive potential who do not plan to use effective contraception.
In females of reproductive potential, pregnancy should be excluded before the initiation of each treatment course of MAVENCLAD and prevented by the use of effective contraception during MAVENCLAD dosing and for at least 6 months after the last dose of each treatment course. Women who become pregnant during treatment with MAVENCLAD should discontinue treatment. Advise women not to breastfeed during dosing with MAVENCLAD and for 10 days after each monthly dosing cycle.
Physicians and patients are encouraged to report pregnancies of women with multiple sclerosis exposed to oral cladribine during pregnancy or within 6 months before conception as well a pregnancies fathered by men with multiple sclerosis who had taken oral cladribine within 6 months before conception by calling EMD Serono's Adverse Event reporting line at 1-800-283-8088 ext. 5563 or by faxing 1-781-681-2961.
Cladribine in animal studies1
There are no adequate data on the developmental risk associated with use of MAVENCLAD in pregnant women.
In preclinical studies:
- Cladribine was embryolethal when administered to pregnant mice and produced malformations in mice and rabbits. The observed developmental effects are consistent with the effects of cladribine on DNA. The amount of cladribine administered to mice was 0, 0.5, 1.5, or 3 mg/kg/d, and the amount administered to rabbits was 0, 0.3, 1, or 3 mg/kg/d‡
- When cladribine was administered by subcutaneous injection to male mice prior to and during mating to untreated females, no effects on fertility were observed. However, an increase in non-motile sperm was observed at the highest dose tested. The amount of cladribine administered to male mice was 0, 1, 5, 10, or 30 mg/kg/d‡
‡The approved dose of MAVENCLAD is a cumulative dose of 3.5 mg/kg over 2 years.
Contraception1
- Effective contraception should be used by females and males during MAVENCLAD dosing and up to 6 months after the last dose in each treatment course
- Because cladribine interferes with DNA synthesis, adverse effects on human gametogenesis could be expected
- Male patients of reproductive potential should take precautions to prevent pregnancy of their partner during MAVENCLAD dosing and for at least 6 months after the last dose in each treatment course
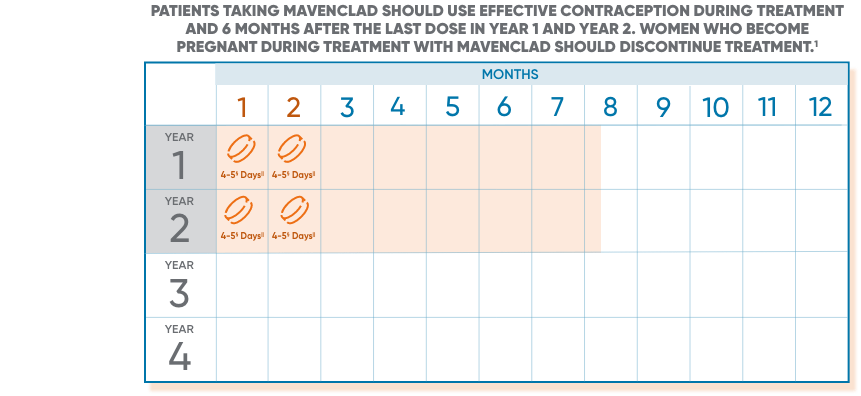
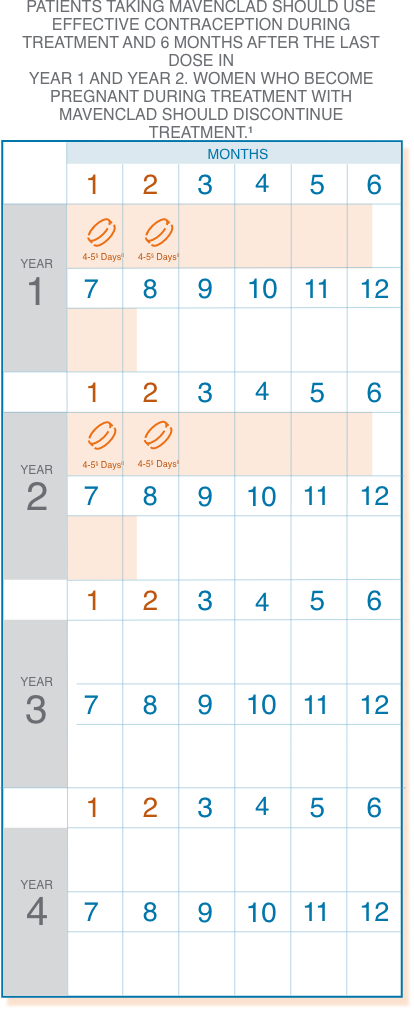
§1 or 2 pills a day depending on weight.
||Treatment days.
Following the administration of 2 treatment courses, do not administer additional MAVENCLAD treatment during the next 2 years. Treatment with MAVENCLAD after 2 years may further increase the risk of malignancy. The safety and efficacy of reinitiating MAVENCLAD more than 2 years after completing 2 treatment courses has not been studied.
Safety and tolerability with MAVENCLAD in CLARITY
The most common (>20%) adverse reactions reported in CLARITY are upper respiratory tract infection, headache, and lymphopenia.1
| Adverse reaction | MAVENCLAD (n=440) % | Placebo (n=435) % |
|---|---|---|
| Upper respiratory tract infection | 38 | 32 |
| Headache | 25 | 19 |
| Lymphopenia | 24 | 2 |
| Nausea | 10 | 9 |
| Back pain | 8 | 6 |
| Arthralgia and arthritis | 7 | 5 |
| Insomnia | 6 | 4 |
| Bronchitis | 5 | 3 |
| Hypertension | 5 | 3 |
| Fever | 5 | 3 |
| Depression | 5 | 3 |
Additional serious adverse reactions and potential risks are discussed below.
Discontinuation rate due to adverse reactions
In the pivotal trial, 3.5% of patients receiving MAVENCLAD discontinued treatment due to adverse reactions vs 2.1% of patients receiving placebo.3
Trial completion
92% of patients treated with MAVENCLAD completed the full 96 weeks of the study vs 87% of patients receiving placebo.1
CLARITY: CLAdRIbine Tablets treating multiple sclerosis orallY.
Lymphopenia
MAVENCLAD causes a dose-dependent reduction in lymphocyte count. In clinical studies, 87% of MAVENCLAD-treated patients experienced lymphopenia. The lowest absolute lymphocyte counts occurred approximately 2 to 3 months after the start of each treatment course and were lower with the second treatment course.1
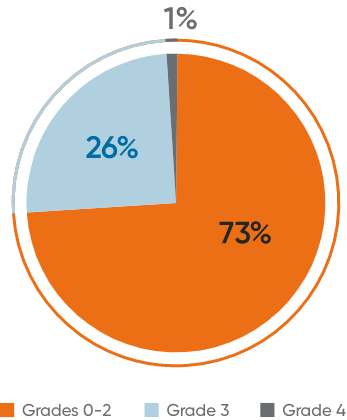
In patients treated with MAVENCLAD 3.5 mg/kg as monotherapy:
- Lymphopenia observed was mostly mild to moderate15
- The majority (73%) of patients who experienced lymphopenia experienced Grade 1 or 21
- 26% experienced Grade 3, and only 1% experienced Grade 41
- 2% of clinical study patients had lymphocyte counts less than 500 cells/μL (severe lymphopenia, Grade 3 or higher) at the end of the second treatment course1
- The median duration of severe lymphopenia was 6.0 weeks to improvement to Grade ≤2 and 28.1 weeks to recovery to Grade ≤110
The incidence of lymphopenia <500 cells/μL was higher in patients who had used drugs to treat relapsing forms of MS prior to study entry (32.1%), compared to those with no prior use of these drugs (23.8%).1
Lymphocytes must be within normal limits before initiating the first treatment course and at least 800 cells/μL before initiating the second treatment course. If necessary, delay the second treatment course for up to 6 months to allow for recovery of lymphocytes to at least 800 cells/μL. If this recovery takes more than 6 months, the patient should not receive further treatment with MAVENCLAD.1
Graded according to the Common Terminology Criteria for Adverse Events (version 5.0). 1, <LLN-800/μL; 2, <800-500/μL; 3, <500-200/μL; 4, <200/μL.16
LLN: lower limit of normal.
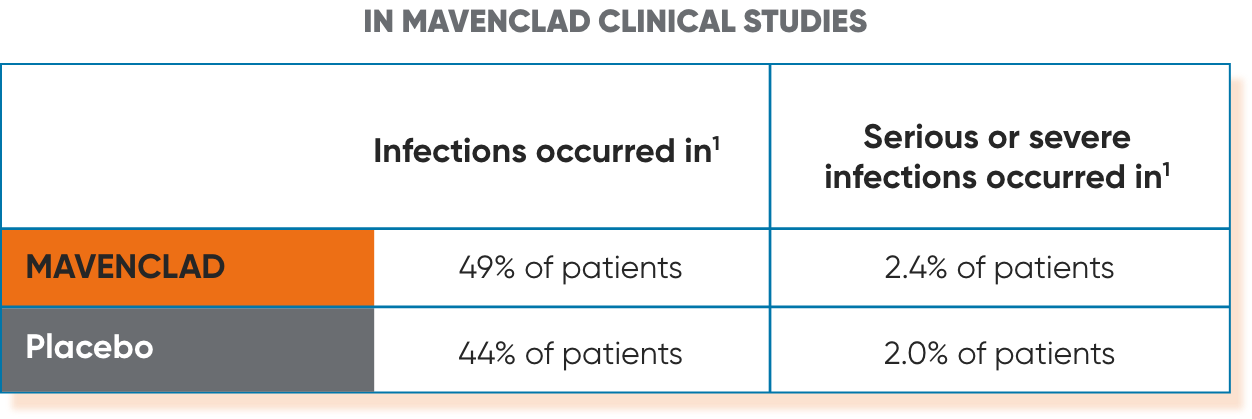
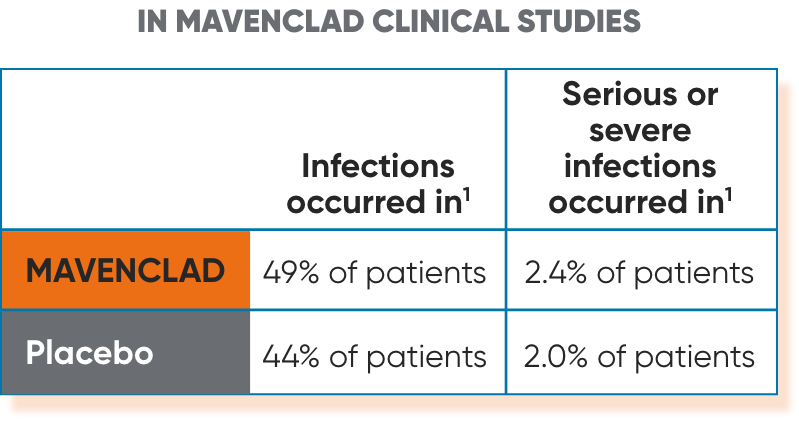
Infections1
Serious, including life-threatening or fatal, infections have occurred. MAVENCLAD reduces the body's immune defense, and an increased risk of infections has been observed in patients receiving MAVENCLAD. The most frequent serious infections included herpes zoster and pyelonephritis. Fungal infections were observed, including cases of coccidioidomycosis.
- Single fatal cases of tuberculosis and fulminant hepatitis B were reported in the clinical program
- HIV infection, active tuberculosis, and active hepatitis must be excluded before initiating MAVENCLAD
- Patients should also be screened for active or latent infections, particularly tuberculosis and hepatitis B and C, before treatment initiation in both treatment years. Delay MAVENCLAD treatment until infection is fully resolved or controlled
- Patients who are seronegative for varicella zoster virus should be vaccinated prior to treatment. Administer live-attenuated or live vaccines at least 4 to 6 weeks prior to starting MAVENCLAD
- Infections that occurred were higher when patients were experiencing Grade 3 and 4 lymphopenia11
INDICATION and IMPORTANT SAFETY INFORMATION for MAVENCLAD® (cladribine) tablets
MAVENCLAD® (cladribine) is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include relapsing-remitting disease and active secondary progressive disease, in adults. Because of its safety profile, use of MAVENCLAD is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS.
Limitations of Use: MAVENCLAD is not recommended for use in patients with clinically isolated syndrome (CIS) because of its safety profile.
IMPORTANT SAFETY INFORMATION
WARNING: MALIGNANCIES and RISK OF TERATOGENICITY
- Treatment with MAVENCLAD may increase the risk of malignancy. MAVENCLAD is contraindicated in patients with current malignancy. In patients with prior malignancy or with increased risk of malignancy, evaluate the benefits and risks of the use of MAVENCLAD on an individual patient basis. Follow standard cancer screening guidelines in patients treated with MAVENCLAD.
- MAVENCLAD is contraindicated for use in pregnant women and in women and men of reproductive potential who do not plan to use effective contraception because of the potential for fetal harm. Malformations and embryolethality occurred in animals. Exclude pregnancy before the start of treatment with MAVENCLAD in females of reproductive potential. Advise females and males of reproductive potential to use effective contraception during MAVENCLAD dosing and for 6 months after the last dose in each treatment course. Stop MAVENCLAD if the patient becomes pregnant.
CONTRAINDICATIONS
- Patients with current malignancy.
- Pregnant women, and women and men of reproductive potential who do not plan to use effective contraception during and for 6 months after the last dose in each treatment course. May cause fetal harm.
- Patients infected with human immunodeficiency virus (HIV).
- Patients with active chronic infections (e.g., hepatitis or tuberculosis).
- Patients with a history of hypersensitivity to cladribine.
- Women intending to breastfeed on a MAVENCLAD treatment day and for 10 days after the last dose.
WARNINGS AND PRECAUTIONS
- Malignancies: Treatment with MAVENCLAD may increase the risk of malignancy. After the completion of 2 treatment courses, do not administer additional MAVENCLAD treatment during the next 2 years. In clinical studies, patients who received additional MAVENCLAD treatment within 2 years after the first 2 treatment courses had an increased incidence of malignancy. The risk of malignancy with reinitiating MAVENCLAD more than 2 years after the completion of 2 treatment courses has not been studied. Follow standard cancer screening guidelines in patients treated with MAVENCLAD.
- Risk of Teratogenicity: MAVENCLAD may cause fetal harm when administered to pregnant women. In females of reproductive potential, exclude pregnancy before initiation of each treatment course of MAVENCLAD and prevent by the use of effective contraception during MAVENCLAD dosing and for at least 6 months after the last dose of each treatment course. Women who become pregnant during treatment with MAVENCLAD should discontinue treatment.
- Lymphopenia: MAVENCLAD causes a dose-dependent reduction in lymphocyte count. Concomitant use of MAVENCLAD with hematotoxic drugs may increase the risk of adverse reactions because of the additive hematological effects. Monitor lymphocyte counts before, during, and after treatment.
- Infections: Serious, including life-threatening or fatal, infections have occurred. MAVENCLAD reduces the body's immune defense, and an increased risk of infections has been observed in patients receiving MAVENCLAD. Infections occurred in 49% of MAVENCLAD-treated patients compared to 44% of patients treated with placebo in clinical studies; serious or severe infections occurred in 2.4% of MAVENCLAD-treated patients and 2.0% of placebo-treated patients. The most frequent serious infections included herpes zoster and pyelonephritis. Fungal infections were observed, including cases of coccidioidomycosis. Single fatal cases of tuberculosis and fulminant hepatitis B were reported in the clinical program.
- Screen patients for active and latent infections (tuberculosis, hepatitis B or C). Delay treatment until infection is fully resolved or controlled.
- Vaccinate patients who are seronegative for varicella zoster virus (VZV) prior to treatment. Vaccinate patients who are seropositive to VZV with recombinant, adjuvanted zoster vaccine either prior to or during treatment, including when their lymphocyte counts are less than or equal to 500 cells per microliter.
- Administer anti-herpes prophylaxis in patients with lymphocyte counts less than 200 cells per microliter. Monitor for infections.
- Progressive multifocal leukoencephalopathy (PML) has been reported in patients treated with parenteral cladribine for oncologic indications. No case of PML has been reported in clinical studies of cladribine in patients with MS. Obtain a baseline magnetic resonance imaging (MRI) within 3 months before initiating the first treatment course of MAVENCLAD. At the first sign of PML, withhold MAVENCLAD and perform an evaluation.
- Administer all immunizations (except as noted for VZV) according to immunization guidelines prior to starting MAVENCLAD. Administer live-attenuated or live vaccines at least 4 to 6 weeks prior to starting MAVENCLAD due to risk of infection.
- Hematologic Toxicity: In addition to lymphopenia, decreases in other blood cells and hematological parameters have been reported with MAVENCLAD in clinical studies. Obtain complete blood count (CBC) with differential including lymphocyte count before and during treatment, periodically thereafter, and when clinically indicated.
- Graft-versus-Host Disease with Blood Transfusions: Transfusion-associated graft-versus-host disease has been observed rarely after transfusion of nonirradiated blood in patients treated with cladribine for non-MS treatment indications. In patients who require blood transfusion, irradiation of cellular blood components is recommended.
- Liver Injury: In clinical studies, 0.3% of MAVENCLAD-treated patients had liver injury (serious or causing treatment discontinuation) compared to 0 placebo patients. Obtain serum aminotransferase, alkaline phosphatase, and total bilirubin levels prior to treatment. Discontinue MAVENCLAD if clinically significant liver injury is suspected.
- Hypersensitivity: If a hypersensitivity reaction is suspected, discontinue MAVENCLAD therapy. Do not use MAVENCLAD in patients with a history of hypersensitivity to cladribine.
- Cardiac Failure: In clinical studies, one MAVENCLAD-treated patient experienced life-threatening acute cardiac failure with myocarditis, which improved after approximately one week. Cases of cardiac failure have also been reported with parenteral cladribine used for treatment indications other than multiple sclerosis. Instruct patients to seek medical advice if they experience symptoms of cardiac failure (e.g., shortness of breath, rapid or irregular heartbeat, swelling).
Adverse Reactions: The most common adverse reactions (incidence of >20%) are upper respiratory tract infection, headache, and lymphopenia.
Drug Interactions: Concomitant use with immunosuppressive or myelosuppressive drugs and some immunomodulatory drugs (e.g., interferon beta) is not recommended and may increase the risk of adverse reactions. Acute short-term therapy with corticosteroids can be administered. Monitor for additive effects on the hematological profile with use of hemotoxic drugs. Avoid concomitant use of antiviral and antiretroviral drugs. Avoid concomitant use of BCRP or ENT/CNT inhibitors as they may alter bioavailability of MAVENCLAD.
Use in Specific Populations: Studies have not been performed in pediatric, or elderly patients >65 years, pregnant or breastfeeding women. Use in patients with moderate to severe renal or hepatic impairment is not recommended.
To report SUSPECTED ADVERSE REACTIONS, contact EMD Serono, Inc. at 1-800-283-8088 ext. 5563 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see FULL PRESCRIBING INFORMATION, including BOXED WARNING.